Theme: Inventing formula for Drug Development and Analytical Techniques
Drug Formulation 2018
We invite all interested participants across the globe to attend 10th Annual Congress on Drug Formulation & Analytical Techniques during December 10-11,2018 in Dubai, UAE which includes prompt keynote presentations, Oral talks, Poster presentations and Exhibitions. Drug Formulation 2018 is a specially designed cluster Pharmaceutical and Medicinal Chemistry Conference. The main theme of this Pharma conferences is “On-going Research & Inventing a Formula for Developing Drug and Analysis” which covers a wide range of critically important sessions.
Working under the theme "Inventing formula for Drug Development and Analytical Techniques" this unique international conference will opportunity to reach the largest assemblage of participants from the Pharmaceutical community to gather and share their insights and convey recent developments in the field of generic drug research and current challenges and possibilities in modeling a new drug and breakthroughs in drug development, Generic drug safety, Novel trends and advanced strategies involving Formulation research. This is a true forum where ideas and discussion is driven by the participants and interaction with peers and others leads to fruitful outcomes.
Drug Formulation 2018 is a 2-day event offering the Exhibition, at venue to showcase the new and emerging technologies and have wider sessions involving Keynote presentation, Oral, YRF ( student presentation), poster, e-poster presentations. World-renowned speakers and eminent delegates across the globe attending the conference, to share their valuable presentation on the most recent and advanced techniques, developments, and the newest updates are the prominent features of the conference.
Why to Attend???
Meet your target audience with members from around the world focused on learning about Pharmaceutical Sciences and Medicinal Chemistry, this is your single best opportunity to reach the largest assemblage of participants from the Pharma and medicinal community, Find out how innovators are closing the gap between product development and adoption, Better identify cyber threats and reduce information security risks, Debate the potential to greater cross-industry and cross-sector collaboration in the race for faster, cheaper, better cures. Conduct demonstrations, distribute information, meet with current and potential Scientists, make a splash with a new research, and receive name recognition at this 2-day event Pharmaceutical Science & Medicinal Chemistry. World-renowned speakers, the most recent techniques, tactics, and the newest updates in Pharmaceutical Sciences fields are hallmarks of this conference.
A Unique Opportunity for Advertisers and Sponsors at this International event:
Target Audience:
- Professors, Associate Professors, Assistant Professors
- PhD Scholars
- Graduates and Post Graduates
- Directors, CEO’s of Organizations
- Association, Association presidents and professionals
- Noble laureates in Health Care and Medicine
- Bio instruments Professionals
- Bio-informatics Professionals
- Software development companies
- Research Institutes and members
- Supply Chain companies
- Manufacturing Companies
- CRO and DATA management Companies
- Training Institutes
- Business Entrepreneur
- Government healthcare departments, HTA’s and drug regulators
- Digital health companies
- Genomics and personalised medicine experts
Track 1: Pharmaceutical Formulations
Pharmaceutical formulation in pharmaceutics is the process in which various chemical substances including the vigorous drug are joined to produce a final medicinal product. Formulation studies involve developing a preparation of the drug which is both stable and acceptable to the patient. For orally taken drugs, this usually contains combining the drug into a tablet or a capsule. Formulations confirm that the drug is compatible with these other substances. In the pharmaceutical drug formulations, the different physical, chemical, and mechanical properties of a drug are considered so as to know what other ingredients should be used in the preparation. The various factors like polymorphism, particle size, pH, and solubility are all considered while formulating the drug, also considering the appearance of the tablet.
Track 2: Formulation R&D
The drug subjected to drug development undergoes number of trials and are screened at different stages to produce a final potent drug intended for the treatment of various diseases. During this process various properties are checked to see whether the drug is non-toxic to living system and is therapeutic or not. Using suitable excipients and technological preparations the active substances are formulated into final dosage form. The last product is the Actual composition of preparation, manufacturing specification. Drug formulation can have a substantial impact on almost every quality characteristic of an API including potency, bioavailability, solubility, dosage, route of administration and stability. In immune offers parenteral drug formulation R&D services for primary stage and preclinical candidates with a special prominence in nanoparticle based drug delivery technology.
Track 3: Clinical Research Vs Clinical Trails
Bioavailability is a subcategory of pharmacological absorption. Bioavailability is generally assessed by finding the area under the plasma concentration–time curve. The factors affecting bioavailability are Pharmaceutics factors, physicochemical properties of drug, Dosage form characteristics & Pharmaceutic Ingredients, Patient related factors like age, Routes of administration (Parenteral, Rectal, Oral, and Topical) etc.
Conducting a BA study enables assessment of the impact of route of administration on BA and defines the absolute BA of the drug released from the drug product. BA for a given formulation provides an assessment of the relative fraction of the orally administered dose that is absorbed into the systemic circulation. If the reference standard is an IV dose, it is referred as Absolute Bioavailability. If the reference standard is any other dosage form than IV it is referred as Relative Bioavailability.
Track 4: Bioavailability Studies and Assessment
Conducting a Bioavailability study enables assessment of the impact of route of administration on BA and defines the absolute bioavailability of the drug released from the drug product. BA for a given formulation provides an assessment of the relative fraction of the orally administered dose that is absorbed into the systemic circulation. If the reference standard is an IV dose, it is referred as Absolute Bioavailability. If the reference standard is any other dosage form than IV, it is referred as Relative Bioavailability. Micro and Micro nutrients play a vital role in bioavailability
Bioavailability is a subcategory of pharmacological absorption. Bioavailability is generally assessed by finding the area under the plasma concentration–time curve. The factors affecting bioavailability are Pharmaceutics factors, physicochemical properties of drug, Dosage form characteristics & Pharmaceutical Ingredients, Patient related factors like age, Routes of administration (Parenteral, Rectal, Oral, and Topical) etc. Topics under this track includes Nutrient Bioavailability, Absolute Bioavailability, Relative Bioavailability, Mineral Bioavailability- Micro and Macro, Vitamins Bioavailability, BA of Contaminants in Soils & Sediments, Drug Absorption and Distribution, Disposition studies, Drug Formulation and Dosage Forms, Product design- Considerations, Bio accessibility Factor.
Track 5: Contract Research Organizations
A contract research organization (CRO) is an organization that provides support to the pharmaceutical, biotechnology, and medical device industries in the form of research services outsourced on a contract basis. A CRO may provide such services as biopharmaceutical development, biologic assay development, commercialization, preclinical research, clinical research, clinical trials management, and pharmacovigilance. CROs also support foundations, research institutions, and universities, in addition to governmental organizations. Many CROs specifically provide clinical-study and clinical-trial support for drugs and/or medical devices. CROs that specialize in clinical-trials services can offer their clients the expertise of moving a new drug or device from its conception to FDA/EMA marketing approval, without the drug sponsor having to maintain a staff for these services.
Track 6: Recent approaches to Biosimilars
The development of biologics calls for overcoming lot many challenges. With initial steps of concepts of biologics, their considerations, essentials for early clinical developments it is very much needed that proper scientific and strategic approaches are taken for the successful development of follow-on-biologics. Moreover, the need for overcoming the challenges continues in the late clinical steps, drug safety factors and labeling requirements. Also, it is much required now to develop a drug product in accordance to quality by design (QbD). This Drug related conference will look at the multiple facets of current challenges in bio similar development and focuses on multiple aspects of bio similar product development to successfully deliver safe, potential and efficacious biologic products to the market
Track 7: Pharmaceutical Freeze Drying Technology
Pharmaceutical freeze Technology is the removal of ice or other frozen solvents from a material through the process of sublimation and the removal of bound water molecules through the process of desorption. Lyophilization and freeze drying are terms that are used interchangeably depending on the industry and location where the drying is taking place. Controlled freeze drying keeps the product temperature low enough during the process to avoid changes in the dried product appearance and characteristics. It is an first-rate method for preserving a wide diversity of heat-sensitive materials such as proteins, microbes, pharmaceuticals, tissues & Plasma
Track 8: Recent advancements in BA/BE Research
The aim of bioavailability study is to find out the dosage form influence on the biological performance of the drug, sensitivity to detect differences in the rate and extent of absorption. Bioavailability and bioequivalence study design involves Single dose or multi dose standard 2x 2 crossovers, Parallel groups, for more than two formulations. Study design meant for estimating essential pharmacokinetic parameters differs significantly from a bioequivalence study meant for comparing the test formulation. The results of a pilot study can be used as the sole basis to document BA or BE provided the study’s design and execution are suitable and enough subjects have completed the study.
Track 9: Bioequivalence Studies and Assessment
Bioequivalence studies are done for Early and late clinical trial formulations, Formulations used in clinical trial and stability studies, if different Clinical trial formulations and to-be-marketed drug product when it comes to cost and productivity metrics, it’s often said that what gets measured gets done. Bioequivalence is determined based on the bioavailability of the innovator medicine versus the generic medicine. A typical outline for a bioequivalence study includes organization of the test and reference items on two events to volunteer subjects, with every organization isolated by a washout period. This Study involves parameters on (Cmax) and (AUC), Statistical evaluation.
Assessment of the bioequivalence of generic versions of certain reference drugs is complicated by the presence of endogenous levels of said compounds which cannot be distinguished from externally derived compound levels following drug administration. If unaccounted for, the presence of endogenous compound biases towards equivalence in bioequivalence studies of these drugs. Bioequivalence assessments may be complicated further as disposition of the exogenous analogue can be subject to various endogenous processes resulting in nonlinear pharmacokinetics. To overcome these inherent biases a number of different strategies have been employed.
Track 10: Nano Medicine & Nano Technology
Nanotechnology is the study of extremely small structures, having size of 0.1 to 100 nm. Nano medicine is a relatively new field of science and technology. Brief explanation of various types of pharmaceutical nano systems is given. Classification of nano materials based on their dimensions is given. An application of Nanotechnology in various fields such as health and medicine, electronics, energy and environment, is discussed in detail. Applications of nano particles in drug delivery, protein and peptide delivery, cancer are explained. Applications of various nano systems in cancer therapy such as carbon nano tube, dendrimers, nano crystal, nano wire, nano shells etc. are given. The advancement in nano technology helps in the treatment of neuro degenerative disorders such as Parkinson’s disease and Alzheimer’s disease. Applications of nano technology in tuberculosis treatment, the clinical application of nanotechnology in operative dentistry, in ophthalmology, in surgery, visualization, tissue engineering, antibiotic resistance, immune response are discussed in this article. Nano pharmaceuticals can be used to detect diseases at much earlier stages.
Track 11: Analytical Method Development and Validation
Analytical method development is the process of choosing an accurate assay procedure to determine the composition of a formulation. It is the process of proving that an analytical method is acceptable for use in laboratory to measure the concentration of subsequent samples Analytical methods should be recycled within GMP and GLP environments and must be developed using the protocols and acceptance conditions set out in the ICH guidelines. Analytical method development and validation stand the continuous and inter-dependent task associated with the research and development, quality control and quality assurance departments. Analytical procedures play a critical role in equivalence and risk assessment, management. It supports in formation of product-specific acceptance criteria and stability of results. Validation should establish that the analytical procedure is suitable for its intended purpose. Design of experiment is a powerful tool for the method characterization and validation.
Track 12: Advances in Chromatography and Mass Spectrometry
Chromatography and mass qualitative analysis is employed for analysis of organic compounds. Electro spray ionization (ESI) could be a method employed in mass spectroscopic analysis. As relate to chromatography and mass spectrometry. HPLC is further flexible informative and trusted by the industry people. Recent advances in sample preparation techniques to beat difficulties encountered throughout measuring of little molecules from bio fluids mistreatment LC-MS. For Measuring, observation and protective your important Investments analytical chemistry instruments are used. Global Bio analysis seminars are conducted and those specifically applied for chromatography assays, ligand binding assays to know more advances.
Track 13: Novel Approaches to Analytical and Bio analytical Methods
Analytical chemistry is that the study of the separation, identification, quality control and quality assurance of the chemical parts of natural and artificial materials .The maintenance of a desired level of quality in an exceptionally package or product, particularly by resources that of attention to every one stage of the method of delivery or production. Bio analytical Chemistry could be a sub-division of Analytical Chemistry that refuges the measuring of medicine, Ion sensors, Proteins and DNA Sequences in unnatural samples or concentrations. Accurate quantification of the drug samples is extremely very important for several scientific endeavors which cannot delay the result. Therefore the Bio analytical Techniques are in the main focused to bring the correct results of the drug sample to supply an ideal result.
Track 14: Latest Drug Development instruments
Our research instruments are designed to assist you in finding highly valuable and rare biological variants among vast cell populations. By increasing speed and reducing cost, our Pico droplet technology can help you save resources whilst boosting your chances of success. Our industrial instruments, our research instruments are semi-automated rather than fully automated. While this means they require slightly more user input to configure, it also means that they are more flexible and can be easily adapted to fit the needs of your unique research project.
Track 15: Role of Chemistry in Pharmaceuticals
Pharmaceutical science is a major field. It involves different areas of science, which are used to bring out pharmaceutical drug compounds. Computational chemistry involved with the design of new chemical compounds as drugs. Pharmaceutical chemistry and Organic chemistry are involved in the preparation of pharmaceutical drugs. Analytical chemistry involves the quality control and analysis of the pharmaceuticals. Pharmaceutics address the problems in making drug formulations like capsules, tablets, injections etc. The area of pharmacology deals with the study of the efficacy and toxicities of newly designed pharmaceutical compounds.
Track 16: Industrial Pharmacy& Physical Pharmacy
Industrial Pharmacy also plays a crucial role in any drug discovery. To any novel drug discovery the industrial approach is very important to get massive commercial application. Few things which have to be considered by industries to provide a safe and cost affective medicine to the patients like Supply chain, Waste management, Product management, Post- marketing surveillance, Good manufacturing practices and Marketing.
The U.S. pharmaceutical market is the world’s most important national market. Together with Canada and Mexico, it represents the largest continental Pharma market worldwide. The United States alone holds some 40 percent of the global pharmaceutical market. In 2014, this share was valued around 365 million U.S. dollars. Many of the global top companies are located in the United States. In 2014, six out of the top eleven companies were U.S.-based.
Track 17: NMR and Analysis of Small Organic Molecules
In bioscience and drugs, to a small grade molecule may be a coffee mass (<900 Daltons [1]) compound which is incapable to facilitate regulate a process, with a size on the order of 10−9 m. Most drug analysis square measure identical little molecules. For analysis of small organic molecules the subsequent devices ought to be recycled are as follows HPLC method, chromatography, Ultraviolet-visible (UV-VIS) spectrophotometry, Infrared (IR) spectrometry and mass spectrometry.
Track 18: Pharma Regulatory affairs
Regulatory affairs (RA), is also called government affairs, is a profession within regulated industries, such as pharmaceuticals, medical devices, energy, banking, telecom etc. Regulatory affairs also has a very specific meaning within the healthcare industries (pharmaceuticals, medical devices, biologics and functional foods)
Regulatory affairs (medical affairs) professionals (aka regulatory professionals) usually have responsibility for the following general areas: Ensuring that their companies comply with all of the regulations and laws pertaining to their business, Working with federal, state and local regulatory agencies and personnel on specific issues affecting their business. i.e. working with such agencies as the Food and Drug Administration or European Medicines Agency(pharmaceuticals and medical devices); The Department of Energy; or the Securities and Exchange Commission(banking), Advising their companies on the regulatory aspects and climate that would affect proposed activities. i.e. describing the "regulatory climate" around issues such as the promotion of prescription drugs and Sarbanes-Oxley compliance.
Relevant Conferences: Drug Formulation Conference | Pharma Meeting | Pharma Events | Drug Research Events | Pharma Conferences
- 5th International Conference on Pain Medicine and Pain Management May 21-22, 2018 | New York, USA
- International Conference on Pharmaceutical Research and Development June 06-07, 2018 Philadelphia, Pennsylvania, USA
- 7th International conference on Pharmaceutical Auditing June 08-09, 2018 Philadelphia, Pennsylvania, USA
- 8th International Conference and Exhibition on Pharmaceutical Regulatory Affairs and IPR June 08-09, 2018 Philadelphia, Pennsylvania, USA
- 4th International Conference on Drug Discovery, Designing and Development June 27-28, 2018 Vancouver, British Columbia, Canada
- International Conference And Exhibition on Drug Safety & Pharmacovigilance August 29-30, 2018 Toronto, Canada
- 3rd International Conference on Generics Drugs and Biosimilars November 15-17, 2018 | Frankfurt, Germany
- 13th International Conference on Biologics and Biosimilars October 24-25, 2018 Boston, USA
- 12th Edition of International Conference on Nanopharmaceutics and Advanced Drug Delivery August 16-17, 2018 ,Dublin, Ireland
- 17th Edition of International Conference and Exhibition on Pharmaceutics & Novel Drug Delivery Systems October 04-06, 2018 ,Moscow, Russia
- 5th NovAliX Conference Biophysics in Drug Discovery June 13-15, 2018, Boston, USA
- 5th Annual Drug Discovery USA Congress 2 - 3 October 2018, San Diego, USA
- 21st Century Drug Discovery and Development for Global Health (S3) October 17—20, 2018 Hotel Palace Berlin, Berlin, Germany
Major Associations:
Drug Information Association | Canadian Society for Pharmaceutical Sciences (CSPS) | Chemical Society Reviews | American Association of Pharmaceutical Scientists (AAPS) | Indonesian pharmaceutical association| Pharmaceutical Association of Malaysia (PhAMA) | Malaysian Pharmaceutical Society | International Society for Biological Therapy of Cancer | Pharmaceutical Group of the European Union (PGEU) | The Pharmaceutical and Novel Drug Delivery Healthcare Sciences Society (PHSS) | Association of the British Pharmaceutical Industry (ABPI) | American Association of Pharmacy Technicians (AAPT) | American Associations of pharmaceutical scientists (AAPS) | The Pharmaceutical Research & Manufacturers Association (PhRMA) | Pharmaceutical Group of European Union (PGEU) |
Related Associations and Societies:
Societies in USA
Drug Information Association | Canadian Society for Pharmaceutical Sciences (CSPS) | Chemical Society Reviews| American Association of Pharmaceutical Scientists (AAPS) | American Association of Pharmacy Technicians (AAPT) | American Associations of pharmaceutical scientists (AAPS) | The Pharmaceutical Research & Manufacturers Association (PhRMA)
Societies in Asia and Pacific
Indonesian pharmaceutical association | Pharmaceutical Association of Malaysia (PhAMA) | Malaysian Pharmaceutical Society | International Society for Biological Therapy of Cancer
Societies in Europe
Pharmaceutical Group of the European Union (PGEU) | The Pharmaceutical and Novel Drug Delivery Healthcare Sciences Society (PHSS) | Association of the British Pharmaceutical Industry (ABPI) | Pharmaceutical Group of European Union (PGEU) | Pharmaceutical Group of European Union (PGEU)
Drug Formulation 2018 provides the best Place to share and gain knowledge. It is going to be a 2 - day event to ensure network for Drug Developing by using the analytical techniques and Formula, global knowledge on pharma, emerging technologies and can transform formulation and drug delivery, Innovative scientific techniques, new utilities of Pharmaceutical Sciences at one place.
Role of Pharma in World:
Pharma companies have always related with consumers. They have marketed directly to users, especially in the U.S., and they involve patients in clinical trials. They thus have some limited experience with consumer-facing business models.In this new era, pharma companies will have to become more proactive with large communities of patients and go beyond lip service in meeting the needs of a wide swath of consumers.
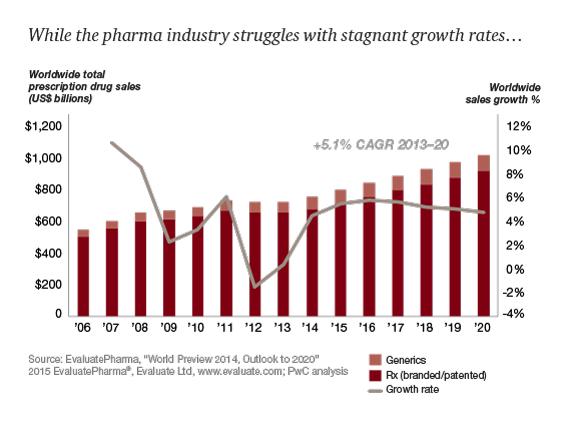
Market Analysis in Arab Countries
Though Arab drug industry is increasingly contributing to the production of biotechnology products, particularly vaccines and herbal medicines, and Chronic drugs. The main challenge is to achieve the regional objective of self-sufficiency in the production of essential drugs and vaccines by using the Analytical techniques and Drug Formulation Methods.
Data from the Union of Arab Manufacturers indicates that the Arab pharmaceutical market was worth US$ 6.20 billion accounting for 1.25 per cent of the global pharmaceutical market in the year 2003. In Arab countries, the local drug production constitutes various percentages of national drug market ranging from 0 per cent up to more than 90 per cent. In average, the local Arab drug industry covers 50 per cent of the total Arab drug market.
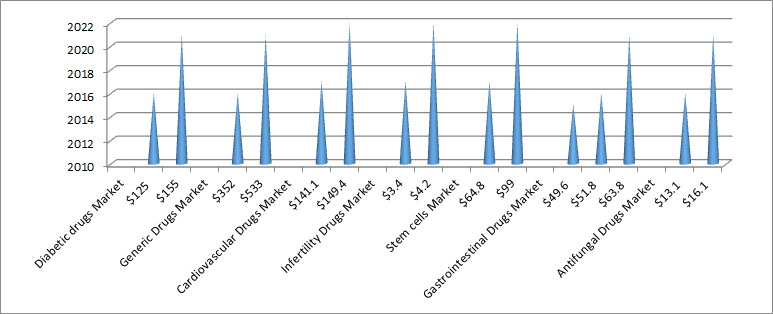
Research and development on - Antifungal Drugs, Diabetes, Generic drugs, Cardiovascular drugs, Infertility drugs, Stem cells in Dubai, UAE
Research and development activities in the Arab pharmaceutical industries focus on the manufacturing process and are often aimed at improving product quality. Small firms may not be involved in research and development activities. Recently, few companies particularly in Jordan and Egypt initiated some innovative research activities based on local medicinal plants.
Recently efforts are being made in Egypt, Saudi Arabia, Jordan and the UAE to start the production of biotechnology products, particularly vaccines and other biological products. The major areas that need to be addressed in order to develop research and development include lack of funding, lack of experienced human resources in certain specialized areas and lack of suitable environment to promote research and development.
Recent Trends in UAE Pharmaceutical Market
The UAE pharmaceutical market was estimated to have sales of around AED 9.6 billion by 2016. These sales are estimated to grow at a CAGR of around 8% and by 2020, the UAE pharmaceutical market is estimated to be worth around AED 13 billion.
Most of the healthcare expenditure in the UAE in 2016 was government expenditure that was estimated to be around AED 42 billion, while private healthcare expenditure was estimated to be around AED 18 billion. Another major trend is the growth of local production of pharmaceuticals which is estimated to grow at a CAGR of around 10% from 2015 to 2018.
The pharmaceutical market in the United Arab Emirates (UAE) is set to grow from just over $3 billion in 2015 to $5.7 billion by 2020, driven by increased healthcare expenditure, mandatory health insurance, and growing medical tourism, according to research and consulting firm Global Data.
Pharmaceutical Companies in UAE:
- Al Razi Pharmacy Company LLC, Abu Dhabi
- Modern Pharmaceutical Co., Abu Dhabi
- Medisal Pharmaceuticals Industry L.L, Abu Dhabi
- Propharma Medical Supplies- Abu Dhabi
- Al Manara Pharmacy - Deerfield Mall, Abu Dhabi
- NeoBioconN FZ LLC, Abu Dhabi
- Boehringer Ingelheim, Dubai
- Novo Nordisk Pharma Gulf Fz Llc, Dubai
- Gulfdrug, Dubai
- Julphar Life, Dubai
- Bayer Middle East FZE, Dubai
- Hoffman La Roche, Dubai
- AK International LLC, Dubai
- IDS - Al Ittihad Drug Store, Dubai
- MEDA Pharmaceuticals, Dubai
- Al Hayat Pharmaceuticals Company, Sharjah
Pharma Societies of Middle East:
- Medicina Group of Pharmacies- Abu Dhabi, Dhubai
- MPC Healthcares- Dubai, Abu Dabi
- Allergan Pharmaceuticals South Africa (Pty) Ltd- Midrand South Africa
- South African Pharmacy Council- Arcadia, SA
- Pharmaceutical Society of Sounth Africa- Lynnwood, SA
- International Society for Pharmaceutical Engineering- Indonesia
- Indonesian Pharmaceutical Association-Indonesia
- Pharmaceutical Association of Malaysia (PhAMA).- Mal
- Malaysian Pharmaceutical Society-Malaysia
- Malaysian Academy of Pharmacy-Malaysia
- The Romanian Society for Pharmaceutical Sciences (SSFR) Romania
- Pharmaceutical group of the European Union- Romania
- Romanian Association of International Drug Manufacturers (ARPIM)-Romania
- European Federation for Pharmaceutical Sciences- Romania
- Pharmaceuticals-Pwc Romania
- The Finnish Pharmaceutical Society- Finland
- The Finnish Society of Physical Pharmacy- Finland
- Pharma Industry Finland (PIF)
Pharma Associations in Middle East:
- The Association of Finnish Pharmacies, Finland
- Polisano Pharmaceuticals, Romania
- The Romanian Association of International Medicines Manufacturers
- Romanian Association of Foreign Drug Manufacturer
- Romanian Association of International Drug Manufacturers
- Indonesian Pharmaceutical Association –GP Farmasi Indonesia
- International Pharmaceutical Manufacturers Group
- FAPA-Indonesian Pharmacist Association
- Indonesian Pharmaceutical Association
- Indonesia Pharma
- PhAMA (Pharmaceutical Association of Malaysia)
- Malaysian Pharmaceutical Society
- PIASA- Pharmaceutical Industry Association of South Africa
- The Innovative Pharmaceutical association South Africa (IPASA)
- FAPMA-The Federation of African Pharmaceutical Manufacturers Associations
- Southern African Generic Medicines AssociationGeneric & Biosimilar Medicines Southern Africa
- Pharmaceutical Manufacturers Association of south Africa
- Clinigen South Africa
- Pharma Solutions LLC- UAE
- Pharmaworld L.L.C.-UAEPharma Service Co., LLC-Dubai, UAE
- Medisal- Pharmaceutical Industry LLC-Abu Dhabi, UAE
Biggest Pharmaceutical Companies in the World:
- Pfizer, The United States
- Novartis, Switzerland
- Merck & Co, The United States
- Sanofi S.A., France
- Roche-, Switzerland
- GlaxoSmithKline, The United Kingdom
- AstraZeneca, The United Kingdom
- Johnson & Johnson, The United States
- Eli Lilly & Co, The United States
- Abbott Laboratories, The United States
- Bristol-Myers Squibb, The United States
- Teva Pharmaceutical Industry, Israel
- Takeda pharma, Japan
- Bayer Schering, Germany
- Boehringer-Ingelheim, Germany
- Astellas, Japan
- Bristol-Myers Squibb, United Kingdom
- Novo Nordisk, Denmark
- Shire, Ireland
- Biogen, United Kingdom
- Daiichi-Sankyo, Japan
- Eisai, Japan
- Otsuka Pharmaceutical, Japan
- Gilead Sciences, The United States
- Mylan, The United States
Conference Highlights
- Pharmaceutical Formulations
- Formulation R&D
- Contract Research Organizations
- Recent approaches to Biosimilars
- Pharmaceutical Freeze Drying Technology
- Recent advancements in BA/BE Research
- Bioequivalence Studies and Assessment
- Nano Medicine & Nano Technology
- Analytical Method Development and Validation
- Advances in Chromatography and Mass Spectrometry
- Novel Approaches to Analytical and Bio analytical Methods
- Latest Drug Development instruments
- Role of Chemistry in Pharmaceuticals
- Industrial Pharmacy& Physical Pharmacy
- NMR and Analysis of Small Organic Molecules
- Bioavailability Studies and Assessment
- Pharma Regulatory affairs
- Clinical Research Vs Clinical Trails
To share your views and research, please click here to register for the Conference.
To Collaborate Scientific Professionals around the World
| Conference Date | December 10-11, 2018 | ||
| Sponsors & Exhibitors |
|
||
| Speaker Opportunity Closed | |||
| Poster Opportunity Closed | Click Here to View | ||
Useful Links
Special Issues
All accepted abstracts will be published in respective Our International Journals.
- Journal of Pharmaceutics & Drug Delivery Research
- Drug Designing: Open Access
- Journal of Developing Drugs
Abstracts will be provided with Digital Object Identifier by


















